
In solid tumors, MRD generally refers to minimal/molecular residual disease, which is identified through tumor-derived molecular abnormalities—most commonly circulating tumor DNA (ctDNA). MRD detection enables the identification of residual disease that cannot be detected by conventional imaging modalities, including PET-CT, or by standard laboratory methods, and reflects the potential persistence of malignancy and risk of subsequent clinical progression.
MRD testing may assist clinicians in earlier identification of patients at high risk of recurrence, thereby providing additional information to support post-treatment clinical decision-making.
In recent years, significant advances have been made in MRD detection technologies for solid tumors. Accumulating evidence from multiple clinical studies conducted worldwide suggests that MRD status is strongly associated with recurrence risk in patients with solid tumors, supporting its potential role in prognostic assessment and in informing individualized treatment strategies.
Addressing the challenges associated with selecting appropriate technical strategies for ctDNA-based MRD detection, Genecast has pioneered an integrated MRD detection solution, Genecast MRD MinerVa Prime, designed to balance the needs of both therapeutic guidance and highly sensitive MRD monitoring.
MinerVa Prime leverages WES Prime, a whole-exome sequencing platform specifically developed for pan–solid tumor applications, to comprehensively identify tumor-specific mutations. Key genes involved in critical oncogenic pathways are sequenced at increased depth, enabling simultaneous support for treatment decision-making and MRD assessment.
Based on the mutation profile identified from tumor tissue, up to 50 tumor-informed variants are selected to construct a personalized ctDNA panel for each patient. cfDNA is then analyzed using ultra-deep sequencing (up to 100,000×), enabling highly sensitive detection of ctDNA at very low allele frequencies.
Analytical performance validation demonstrates that Genecast MinerVa Prime achieves a limit of detection as low as 0.0025%, substantially exceeding the sensitivity threshold of 0.02% recommended in the Chinese Expert Consensus on MRD Detection and Clinical Application in Lung Cancer. The assay also shows consistently high specificity, exceeding 99.5%, supporting reliable MRD detection in clinical settings.
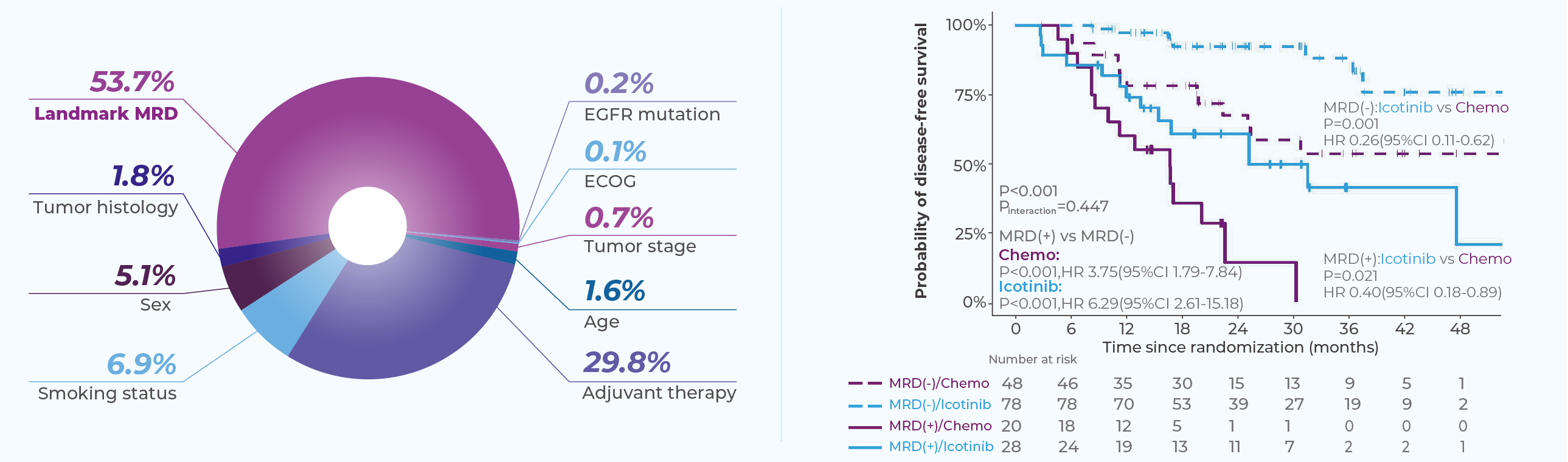
Genecast MRD MinerVa Prime has been evaluated in the EVIDENCE cohort, the large-scale, randomized phase III adjuvant clinical trial conducted in a Chinese population of EGFR-positive non–small cell lung cancer patients.
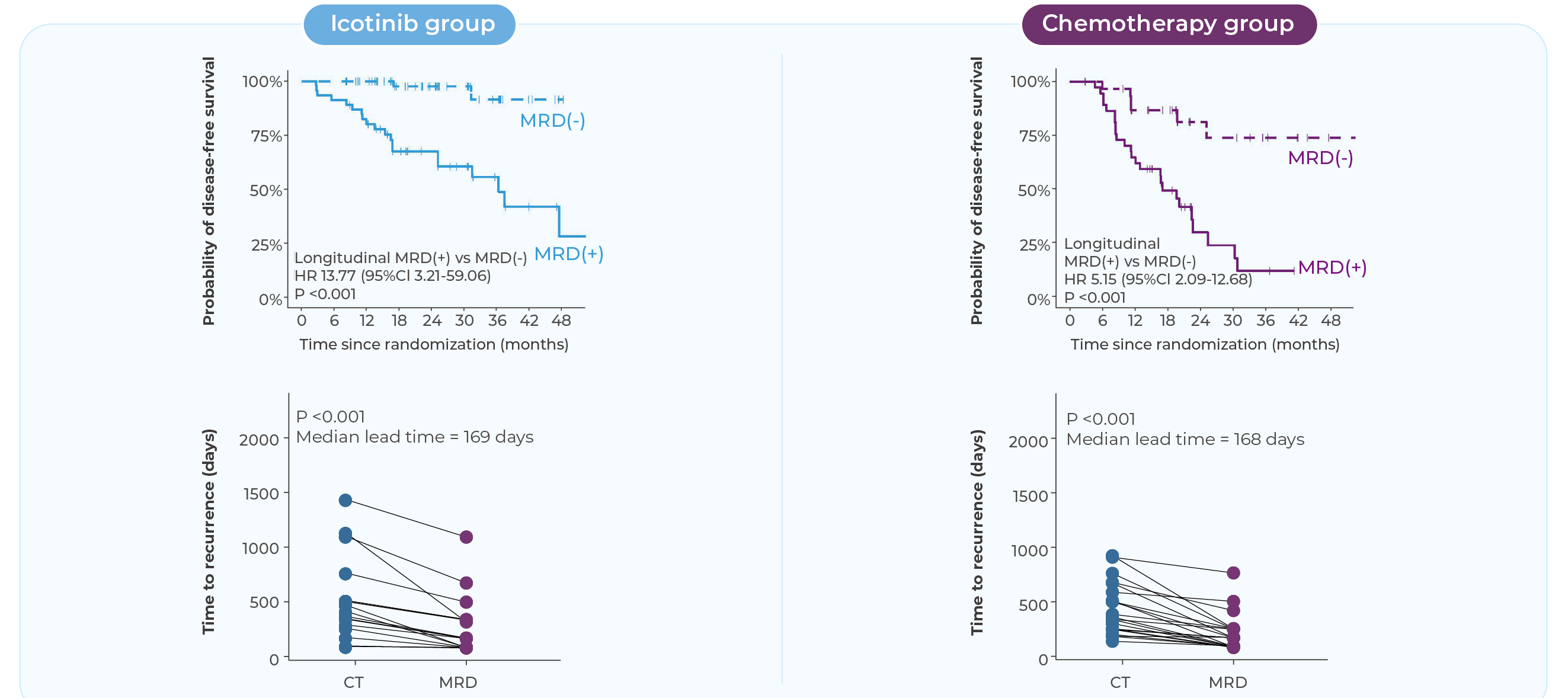
Interim results from this cohort were presented orally at the 2024 World Conference on Lung Cancer. The findings demonstrated that longitudinal ctDNA-based MRD monitoring showed strong predictive value for disease recurrence, with a negative predictive value of 91.3% within the study population. In addition, MRD positivity preceded radiographic evidence of recurrence by a median of 168 days, highlighting the potential of MRD monitoring to enable earlier detection of relapse compared with conventional imaging.
| Enrichment Method | Hybrid capture | |
| Panel Design | Tumor informed WES + personalized 50 and core sites | |
| Sequencing Platforms | NovaSeq 6000/DNBSEQ-T7/DNBSEQ-G400 | |
| Package | Tissue test: 24 tests/kit | Custom test: 24 tests/kit |
| Code | ZYCE-00401 | |
 Prognostic Stratification
Prognostic Stratification
High-sensitivity ctDNA-based MRD testing enables early identification of patients at increased risk of recurrence after surgery or systemic therapy, providing additional molecular-level prognostic information beyond conventional clinical risk factors.
 Longitudinal Monitoring
Longitudinal Monitoring
Serial MRD testing supports longitudinal monitoring of molecular disease dynamics. MRD positivity has been shown to precede radiographic recurrence by several months, allowing earlier awareness of potential disease progression.
 Guiding Treatment Decisions
Guiding Treatment Decisions
Emerging evidence suggests that MRD status may support individualized adjuvant treatment strategies after curative-intent therapy. ctDNA-guided approaches have demonstrated the potential to optimize treatment intensity in selected patient populations.
 Response Evaluation
Response Evaluation
During systemic therapy, MRD serves as a sensitive marker for dynamic response assessment. Changes in MRD status may help identify treatment benefit or emerging resistance and support more personalized long-term management.

Tumor cellularity ≥ 20%;
For tissue areas ≥ 1 cm², provide 12 wax rolls or unstained slides (5–10 μm each).
For tissue areas ranging from 0.1 to 1 cm², or for biopsy (needle-puncture) samples, provide 15 wax rolls or unstained slides (5–10 μm each).
cfDNA or EDTA BCT: 1 tube (≥ 8 mL whole blood)
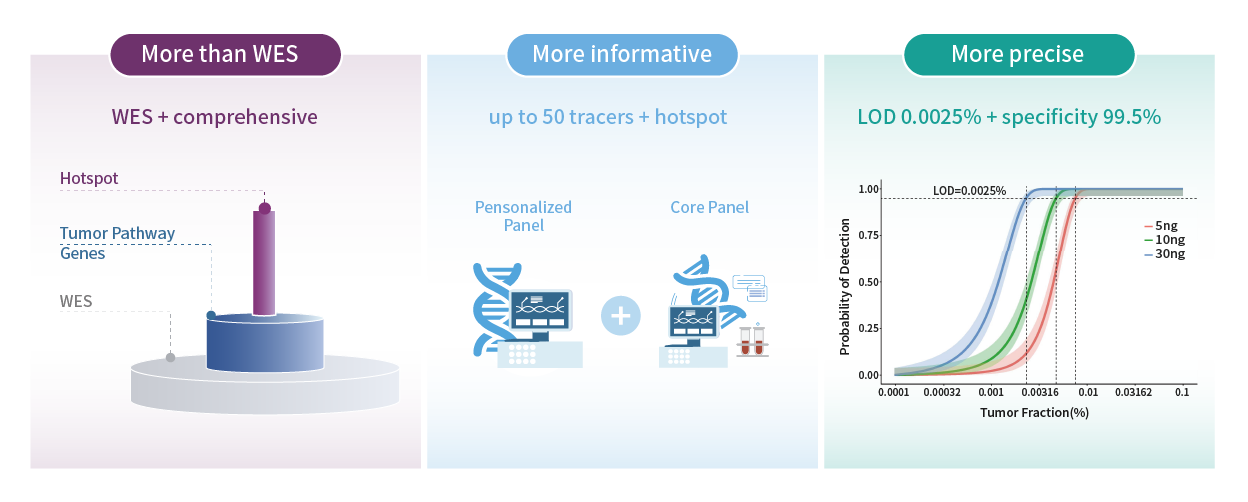
MinerVa Prime employs a proprietary variant prioritization scoring algorithm to select up to 50 high-confidence tumor-specific variants for the construction of a personalized MRD panel. The customized panel design and manufacturing process is highly robust, achieving a 100% customization success rate in the EVIDENCE study.
MRD testing utilizes a dual-panel strategy: while tracking patient-specific tissue-informed variants, it simultaneously interrogates over 1,000 predefined, high-evidence tumor-related hotspot mutations to ensure detection of clinically relevant de novo mutations in plasma.
Ultra-high sequencing depth of 100,000× , combined with multi-variant joint confidence calling, enables highly sensitive MRD detection with a limit of detection as low as 0.0025%. By reducing background noise across the molecular, variant, and sample levels, analytical validation demonstrates consistently high specificity exceeding 99.5%.
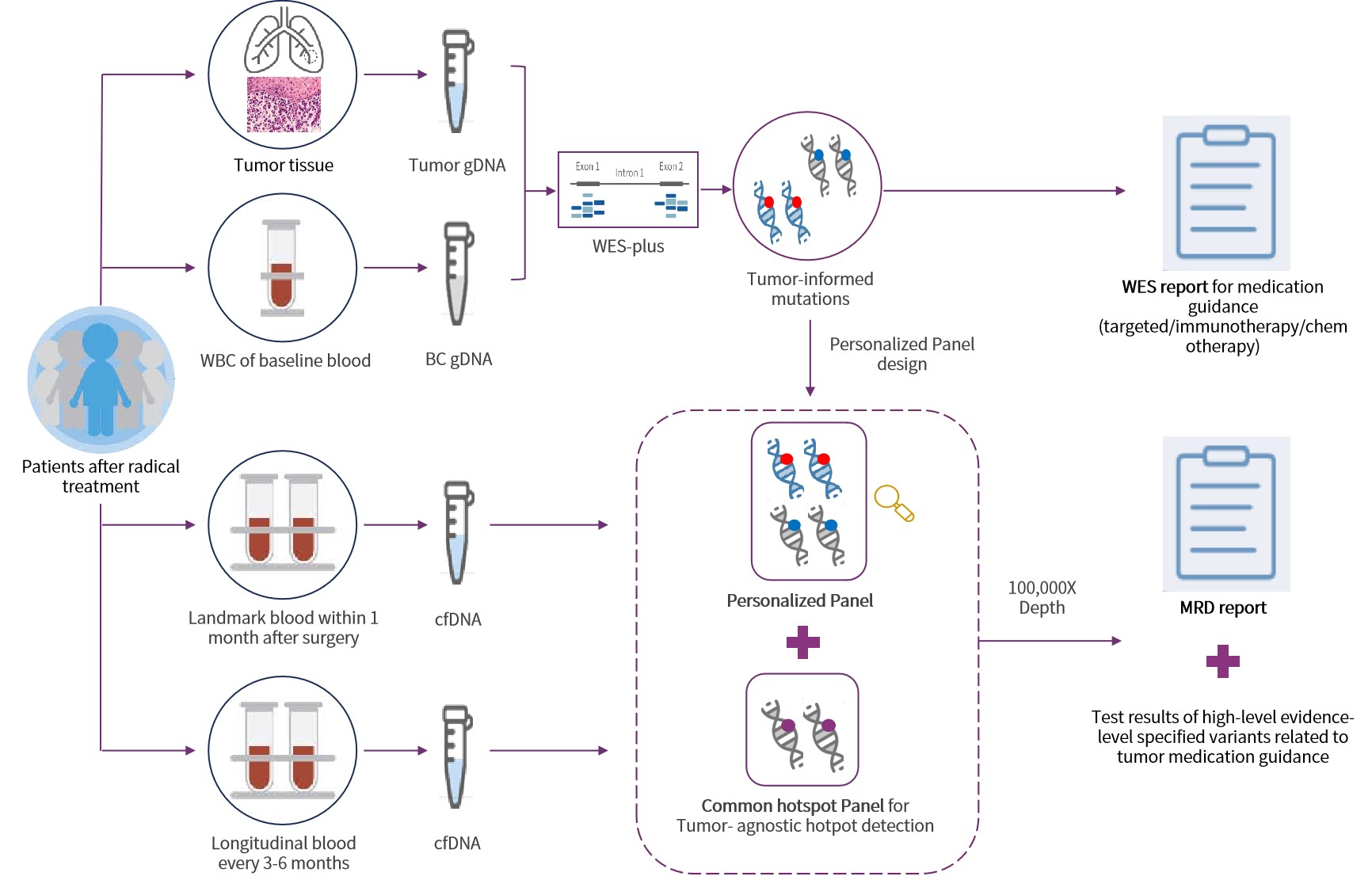

WES Prime design for MRD

99.5% specificity and 0.0025% LoD
Multidimensional noise feature characterization

91.3% NPV, Leading time168 days