
Cancer treatment has evolved from traditional chemotherapy and radiation to the era of personalized or precision medicine. In this journey, diagnosis leads the way: genetic testing now plays a key role in guiding personalized treatment. It helps determine tumor subtypes, informs treatment selection, assesses hereditary risk, and predicts prognosis and recurrence.
NGS enables comprehensive testing in a single run. It is especially useful when tissue samples are limited or when complex mutations are involved, and today it is widely adopted in clinical practice.
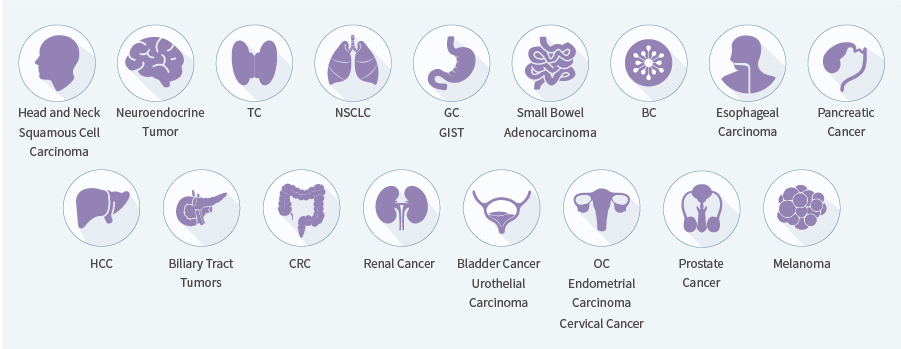
Genecast OncoDuo Prime is a precision therapy test for patients with 20+ solid tumor types. It combines DNA and RNA-based NGS to detect 4 major genomic alterations—SNV, InDel, CNV, and fusions—providing information to guide treatment selection and prognosis.
The test also includes MSI analysis, offering evidence to support the use of immune checkpoint inhibitors and enabling more comprehensive, personalized cancer care.
| Enrichment Method | Hybrid capture | |
| Panel Design | 219 genes + MSI, DNA 188 genes + RNA 52 genes | |
| Sequencing Platforms | DNBSEQ-G99/DNBSEQ-G400/DNBSEQ-T7/NextSeq 550/NovaSeq6000 | |
| Package | 24 tests/kit | |
| Catalog Number | ZYFR-02001 | |
 Therapy Selection
Therapy Selection
Guiding treatment decisions for targeted therapy, immunotherapy, and chemotherapy.
 Prognosis/Diagnosis
Prognosis/Diagnosis
Assisting in prognosis assessment and providing diagnostic information.
 Supports (Neo)Adjuvant and Systemic Therapy Decisions
Supports (Neo)Adjuvant and Systemic Therapy Decisions
Detects SNVs, CNVs, and InDels across 188 genes at the DNA level, and 52 clinically relevant fusions at the RNA level, enabling personalized treatment planning for both adjuvant and systemic settings.

| Type | Requirements | Sample Quantity |
| Surgical/Large Biopsy Tissue Sections | Area ≥ 0.5 cm2, Thickness 5-10 μm; Tumor cell content ≥ 10%; Storage period ≤ 3 years. | ≥ 10 unstained slides/Wax rolls with 4 slices |
| Small Biopsy Tissue Sections | Area ≤ 0.5 cm2, Thickness 5-10 μm; Tumor cell content ≥10%; Storage period ≤ 3 years. | ≥ 15 unstained slides/Wax rolls with 6 slices |

DNA+RNA dual-assay

Target 219 clinical actionable genes and MSI

Assay success rate >95%; accuracy >99%

Compatible with MGI and Illumina sequencing platforms