
In solid tumors, MRD generally refers to minimal/molecular residual disease, which is identified through tumor-derived molecular abnormalities—most commonly circulating tumor DNA (ctDNA). MRD detection enables the identification of residual disease that cannot be detected by conventional imaging modalities, including PET-CT, or by standard laboratory methods, and reflects the potential persistence of malignancy and risk of subsequent clinical progression.
MRD testing may assist clinicians in earlier identification of patients at high risk of recurrence, thereby providing additional information to support post-treatment clinical decision-making.
In recent years, significant advances have been made in MRD detection technologies for solid tumors. Accumulating evidence from multiple clinical studies conducted worldwide suggests that MRD status is strongly associated with recurrence risk in patients with solid tumors, supporting its potential role in prognostic assessment and in informing individualized treatment strategies.
Genecast MRD MinerVa is a tumor-informed ctDNA assay that relies on tumor-specific mutations as prior knowledge for ctDNA detection. It begins by testing the patient's tumor tissue using a fixed panel of 769 genes with a high-depth sequencing coverage of 30,000X to establish the personalized tumor mutation profile. Following this, it examines the existence of ctDNA mutations within this profile in the plasma by utilizing the panel of 769 genes with differential coverage weights.
Genecast MRD MinerVa has been evaluated in the LUNGCA cohort, the most large-scale, prospective, multi-center clinical trial conducted in a Chinese population of stage I-III non–small cell lung cancer patients.
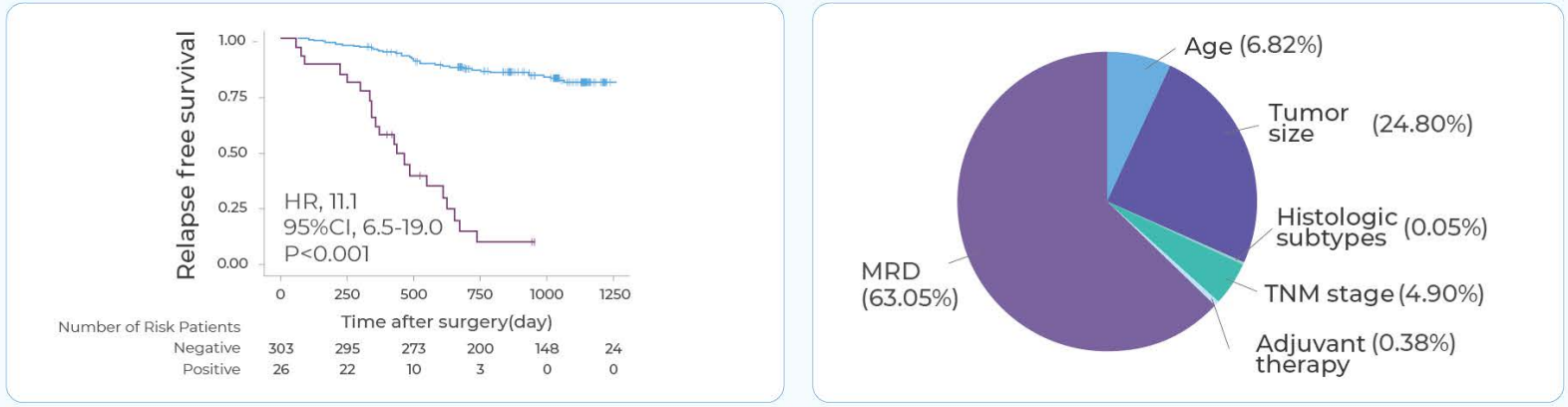
It confirmed that detecting ctDNA-MRD post-surgery strongly predicts recurrence—more strongly than traditional clinical factors like stage. Serial ctDNA-MRD monitoring can detect recurrence a median of 268 days earlier than imaging. The study also showed that MRD-positive patients benefit significantly from adjuvant therapy, while MRD-negative patients may not. These results validate the MinerVa assay as an accurate and reliable tool for early MRD detection and postoperative decision-making.
| Enrichment Method | Hybrid capture | |
| Panel Design | Tumor informed Tissue 769 gene panel+ plasma tracking | |
| Sequencing Platforms | NovaSeq 6000/DNBSEQ-T7/DNBSEQ-G400 | |
| Package | Tissue test: 24 tests/kit | Plasma test: 24 tests/kit |
| Catalog Number | ZYCE-00601 | |
 Prognostic Stratification
Prognostic Stratification
High-sensitivity ctDNA-based MRD testing identifies patients at higher risk of recurrence after surgery or therapy, offering molecular-level insight beyond standard clinical factors.
 Longitudinal Monitoring for Recurrence
Longitudinal Monitoring for Recurrence
Serial MRD testing tracks molecular disease dynamics. A positive result can signal recurrence months before it appears on scans, allowing for earlier clinical awareness.
 Response Evaluation & Guiding Therapy
Response Evaluation & Guiding Therapy
During treatment, changes in MRD status act as a sensitive marker for response or emerging resistance. This supports personalized management, and growing evidence shows MRD can help guide adjuvant therapy decisions and optimize treatment intensity.

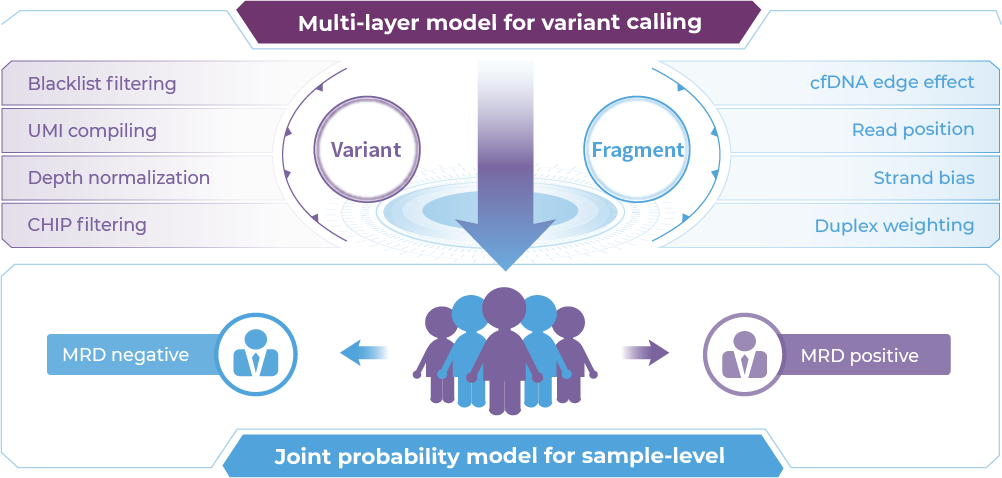

Fixed panel design for MRD

≥99.5% specificity and 0.007% LoD

Multi-layer model for variant calling

91.5%NPV, leading time 268 days